Supporting the safe use of monitored drugs
The New Brunswick Prescription Monitoring Program (PMP), aims to reduce prescription drug misuse and diversion, support safer and coordinated patient care, and promote appropriate prescribing practices. Authorized health-care providers can access and use PMP information to make safer, more informed decisions about patient care.
Information on Prescription Monitoring Program
Does NB currently have a Prescription Monitoring Program?
The Department of Health operates a Prescription Monitoring Program (PMP), a system that tracks prescriptions of monitored drugs dispensed at community pharmacies in New Brunswick. Monitored drugs include opioids such as Dilaudid and Percocet, and controlled drugs such as Ritalin and Ativan. Health professionals can access PMP information through the electronic health record to help identify individuals at risk of harm associated with monitored drugs.
The importance of the PMP has increased in the last few years with the ongoing challenges associated with the growing misuse of medications for pain management and misuse of stimulants.
Why is a new Prescription Monitoring Program system required?
We recognized the need to improve the current system and to take advantage of new technology. The new PMP system has enhanced functionality to better support patient care and promote optimal prescribing and use of drugs prone to misuse.
Feedback received through consultations on the operational review of the Prescription Monitoring Act highlighted the need to improve the system to effectively support clinical decision making. The report on the review presents stakeholder input and an Action Plan to transform the PMP into an effective, adaptive, robust, and resourceful tool.
https://www.legnb.ca/content/house_business/60/1/tabled_documents/5/prescription-monitoring-act.pdf (May 2021)
What is MaveRx?
MaveRxNB is the New Brunswick Prescription Monitoring Program (PMP). The legislation for the PMP, the Prescription Monitoring Act, has existed since 2009. The initial PMP platform was available since 2018 within the Electronic Health Record (EHR). The modernized prescription monitoring platform is rich and intuitive features to aid public health and clinical stakeholders in the fight against prescription drug abuse, opioid addiction, and substance use disorder. Plans are underway to integrate the system into the EHR and other health technologies to improve the user experience.
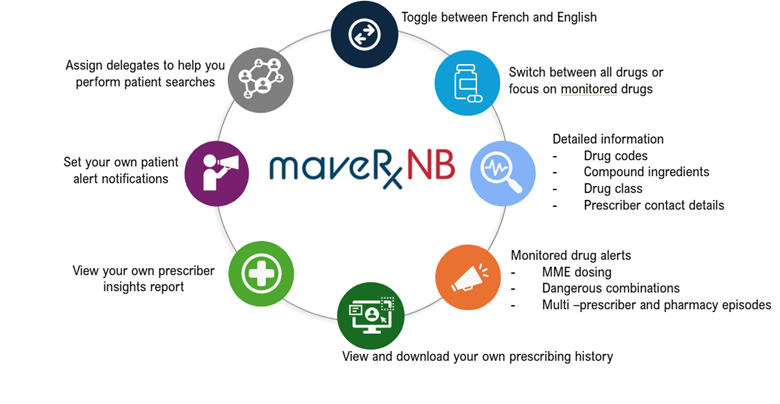
Figure 1: Key Features of the Modernized PMP System
Is prescription drug abuse a problem in New Brunswick?
Misuse of prescription opioids and stimulants is a serious public health problem in all provinces and territories in Canada, including New Brunswick.
The opioid overdose crisis has continued to worsen significantly during the course of the COVID-19 pandemic. The Public Health Agency of Canada (PHAC) reports that 2020 was the worst year for overdoses and deaths in Canada since they started to monitor the crisis in 2016. In New Brunswick, there were four times more overdose deaths than COVID-related deaths in 2020. In 2024, there were 1.9x more opioids related death compared to 2020.
Mandatory Registration and Use of the PMP
Effective August 31, 2026, all prescribers are required to register to the new PMP, MaveRxNB (pronounced “Mavericks NB”), and review the information in the PMP before prescribing or dispensing monitored drugs for a patient.
Prescribers include physicians, dentists, nurse practitioners, optometrists, podiatrists, midwives, and pharmacists. MaveRxNB features risk indicators, visual analytical tools, and prescriber-set notifications that help clinicians assess potential harm to a patient and manage their progress on medication treatments.
For example, prescribers and pharmacists can assess risk of harm due to:
- Potentially high doses and long-term use
- Multiple prescriptions for similar monitored drugs
- Different pharmacies where prescriptions have been filled
- Possible risky combinations of monitored drugs
Health-care providers can also draw insights into their prescribing practices with their own individualized prescriber reports that allow them to assess how their prescribing may affect their patients based on multiple risk metrics. MaveRxNB also supports a busy health-care provider’s workflow by allowing their team to act as delegates to access the system and search for patients on their behalf.
MaveRx Registration and Training
Access to the new PMP requires users to register into the system. To register, enroll in training, or learn more about the new PMP, prescribers are encouraged to contact the PMP support team at pmp.psp@gnb.ca or 506 444-4932 (Monday to Friday, 8:30 AM to 4:00 PM, excluding holidays).
Monitored Drug List
The Monitored Drug List is now available on the GNB website. Any changes to the Monitored Drug List will result in a revised list that will be published on the website.
Frequently Asked Questions
Q: Who is required to register?
A: All prescribers within New Brunswick with an active license are required to register, excluding veterinarians. This includes physicians, physician assistants, dentists, pharmacists, nurse practitioners, optometrists, midwives, and podiatrists. It is voluntary for paramedics and delegates to register and use the system. Delegates are individuals who are authorized to perform patient searches on behalf of a prescriber. They include nurses, administrative assistants, pharmacy technicians, and dental hygienists.
Q: When am I required to use the system?
A: Prescribers are required to review the information within the PMP prior to prescribing or dispensing a monitored drug. A list of monitored drugs can be found on the government website.
Q: How do I register on MaveRx?
A: Please contact the PMP support team at pmp.psp@gnb.ca or 506 444-4932 (Monday to Friday, 8:30 AM to 4:00 PM, excluding holidays).
Q: I recently completed privacy training within the Regional Health Authorities. Do I need to complete your privacy training?
A: No, privacy training within Regional Health Authorities will be accepted in lieu of our privacy training. You will be required to provide an attestation.
Q: I do not work with Regional Health Authorities, but I recently completed privacy training within my organization. Do I need to complete yours?
A: Please send the privacy training certificate to the PMP support team to review. We will confirm if it will be accepted in lieu of ours.
Q: Can I complete the privacy training online?
A: Yes, you can complete the required privacy training online. Please contact us.
Q: How would I know the drugs currently on the monitored list?
A: The monitored drug list is published here: NB PMP website.
Contributed by the Department of Health




